Kinases are the major anticancer drug target class of the 21st century1 with nearly 60 small molecule kinase inhibitors approved for clinical use in the first two decades. Key to the success of kinase inhibitor therapy has been the simultaneous development of biomarker assays to enable the selection of patients most likely to respond in the clinic.
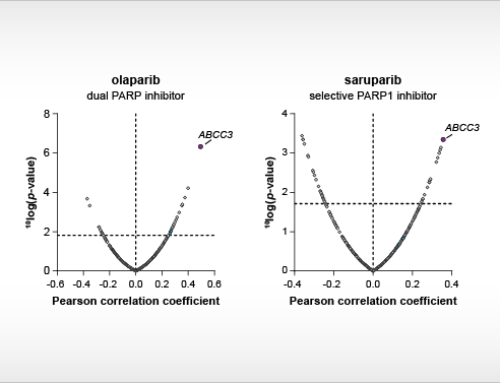
Cancer cell panel profiling is the parallel testing of compounds on a large panel of cancer cell lines. By correlating drug sensitivity with genomic information of the cell lines, novel predictive drug response biomarkers can be identified.
In this webinar, Dr. Guido Zaman (Managing Director & Head of Biology) and Jeffrey Kooijman (Bioinformatician) of NTRC, present the cancer cell panel and biochemical profiling of twenty kinase inhibitors approved for clinical use in cancer therapy between 2018 and 2020. The data presented today builds on the previous work that NTRC scientists have done for all FDA-approved kinase inhibitors up to 2018.2,3 This webinar focuses on the comparison of cellular and biochemical inhibition profiles of the newly approved kinase inhibitors and those of previously approved or clinical stage competitors. In addition, the speakers present how predictive drug response markers were identified for patient stratification and drug repurposing.
What You Will Learn:
- Biochemical kinase profiling and cancer cell panel profiling and how they compare
- How to identify predictive drug response biomarkers by cancer cell panel profiling
- Comparative cancer cell panel profiling of different kinase inhibitors binding to the same target
Speakers

Dr. Guido Zaman, Head of Biology, NTRC

Jeffrey Kooijman, Bioinformatician, NTRC
Access the Webinar
References
- Cohen P. Protein kinases — The major drug targets of the 21st century? Nat Rev Drug Discov. 2002;1:309-15.
- Uitdehaag JCM, de Roos JADM, van Doornmalen AM, Prinsen MBW, de Man J, Tanizawa Y, et al. Comparison of the cancer gene targeting and biochemical selectivities of all targeted kinase inhibitors approved for clinical use. PLoS ONE. 2014;9:e92146.
- Uitdehaag JCM, Kooijman JJ, de Roos JADM, Prinsen MBW, Dylus J, Willemsen-Seegers N, et al. Combined cellular and biochemical profiling to identify drug response biomarkers for kinase inhibitors approved for clinical use between 2013 and 2017. Mol Cancer Ther. 2019;18:470-81.