Cell Proliferation Assays Using High-Throughput Screening
Screen for Drug Efficacy with Reproducible Cellular Compound Profiling Services
Proliferation assays in cancer cell lines are widely applied in drug discovery projects to screen drug efficacy. Screens may cover a large cancer cell line panel to obtain an unbiased view of your compound’s activity and selectivity. Studies can also be run on a selection of cell lines, for instance cell lines that are known for high expression of your target. Dose response curves are generated on the basis of nine dilution points with duplicate measurements. Activity screening of early hits (‘on/off screening’) is done via three dilution points.
Cancer Cell Panel Profiling via Oncolines®
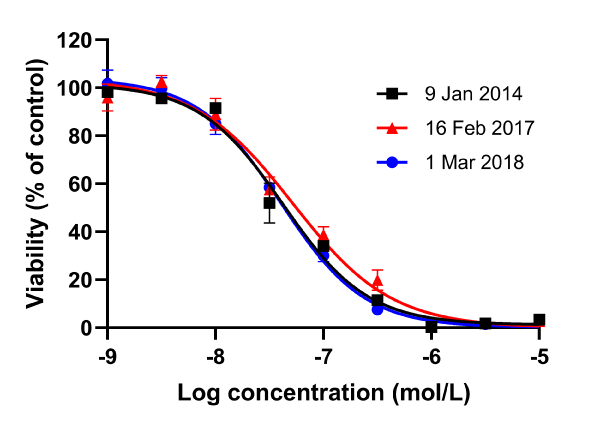
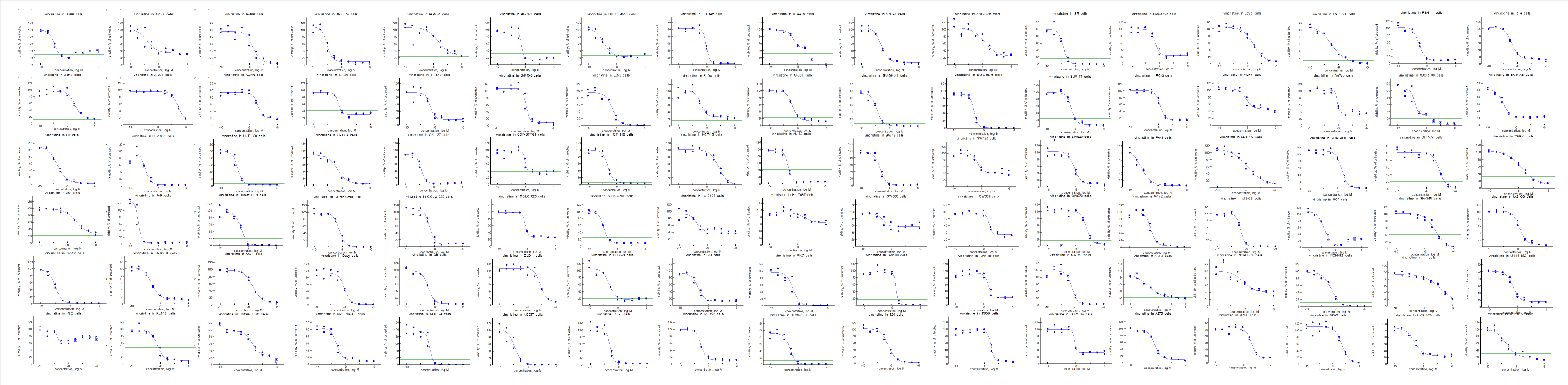
Oncolines® are proliferation assays with a panel of 102 cancer cell lines covering a diverse set of tissue origins. All cell lines have been genetically characterized for mutations and gene expression, and stocks of low passages are used in the screens to prevent genetic drifting. The quality features result in highly reproducible data. The doubling time of each cell line is monitored in every study. As controls, we always include untreated wells in the studies, as well as the reference compound doxorubicin. Bi-phasic behaviour is recognised by visual inspection of the curves. We do not extrapolate dose response curves for the determination of IC50 values. In case the potency of your compound falls outside the measured range, we will retest the compound in an adapted concentration range to accurately determine the growth parameters.
Oncolines® delivers 102 dose response curves in case of full panel testing. The response parameters can be used in bioinformatics studies with the aim finding a mechanistic hypothesis before entering the clinic.
The incubation times for Oncolines® profiling are optimized for three and five days and media are used as recommended by the original investigator for each cell line. To optimally benefit from bioinformatics analysis for biomarker discovery we recommend to run the full panel. If you are more interested in a subset of cell lines, we can also suit your needs.
We have a commercial licence from the ATCC for all the cell lines in the Oncolines® panel.
Biomarker Analysis, Tissue Sensitivity and Targeting
The Oncolines® response parameters can be used to investigate the mechanism of action of your compound without the need for additional experiments. For instance, we perform ANOVA analysis on IC50 values and gene mutation data to identify genomic drug response biomarkers for cancer. Case studies have confirmed patient stratification markers that have been used in the clinic (Kooijman et al., 2022; Uitdehaag et al., 2019).